Particles in biopharmaceuticals (subvisible and visible particulates) can impact drug safety and efficacy, making them a critical quality attribute. The PartiGen Particle Analyzer, leveraging microscopic imaging and Raman spectroscopic detection, enables rapid and accurate characterization, identification, and traceability of particulates. It provides robust data support for formulation development, process optimization, stability studies, manufacturing deviation investigations, and quality control of antibody-based drugs, cell and gene therapies, and biologic vaccines.
-
Microscopic Counting & Particle Size Analysis

-
Spectroscopic Identification & Traceability

-
Pharmaceutical Specific Database

-
Applicable to a wide range of dosage forms


Detection Workflow for Insoluble Particles in Biologics
When insoluble particles are observed under optical inspection, the sample can be directly applied to the XtremSig-P Particle Detection Chip without complex pre-treatment. The PartiGen Particle Analyzer then performs measurements following the workflow:
1. Wide-Field Scanning & Localization – Identifies and maps particles across a broad field of view.
2. Image Analysis – Analyzes particle morphology for size and shape characterization.
3. Spectroscopic Detection – Captures Raman spectra for precise chemical identification.
4. Database Search – Matches spectral data with the pharmaceutical core database for traceability.
This streamlined approach provides comprehensive insights into particle morphology and composition, ensuring reliable data for formulation quality assessment.
Advanced Data Analysis for Particulate Matter with Integrated Algorithms

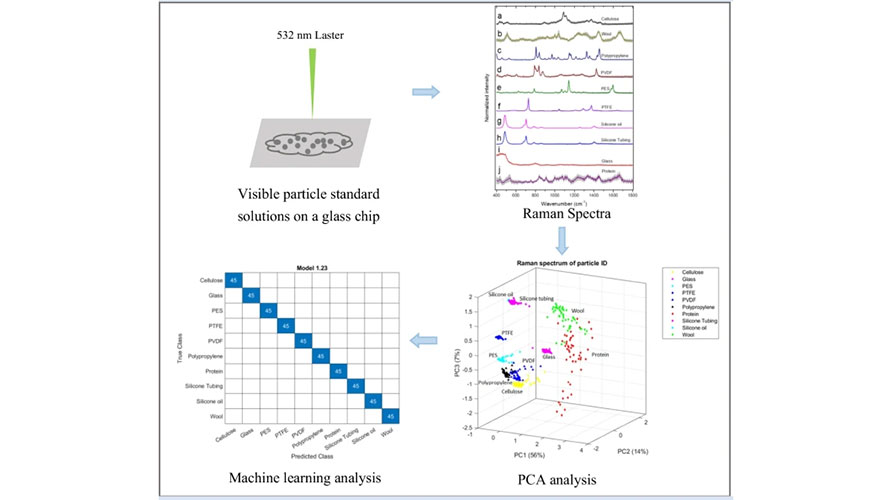
After collecting data on insoluble particles in formulations using the PartiGen Particle Analyzer, intelligent algorithm process the data to generate clear and intuitive visual reports. Additionally, the software integrates multiple spectral analysis algorithms to extract deeper insights, such as:
• Tracking changes in particle size distribution over time or across different production batches.
• Identifying correlations between particle composition and formulation stability.
These capabilities support comprehensive particle characterization, process optimization, and quality control in pharmaceutical development.
Featured Products
-

 AAPS PHARMSCITECH丨Visible Particle Identifcation Using Raman Spectroscopy and Machine Learning2024.03.16
AAPS PHARMSCITECH丨Visible Particle Identifcation Using Raman Spectroscopy and Machine Learning2024.03.16













